Photons as light quanta
A photon is a quantum of light. Our picture of light, to this point, has been that of a wave. Wave-like characteristics are responsible for diffraction and refraction. However, light is absorbed and emitted one photon at a time. The energy of a photon is related to the frequency of the light wave by Planck's constant:
![]()
Photons are massless particles. The idea of massless particles may seem a bit strange. To understand them better, we consider the expressions for energy and momentum of particles of mass m.
![]()
Massless particles can be thought of as the limit of massive particles with m approaching zero. Even though the mass may be infinitesimally small, the factor g may approach infinity as the velocity approaches c. Thus for massless particles, the above expressions are meaningless, but the ratio of the two expressions is valid as the factors g and m cancel.
![]()
We have made use of the fact that the velocity v equals c for massless particles. Using the relation between wavelength and frequency, c = l f, one gets three useful expressions relating energy, momentum, frequency and wavelength.
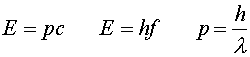
Thus light of shorter wavelengths interacts in the form of photons with higher energy than light with longer wavelengths. We will discuss later the fact that the last of these three formulas is also true for particles with non-zero mass.