PHY919: Modern Electronic Structure Theory
Michigan State University, Fall Semester 2017
Homework #6
Due date: October 10, 2017
This homework will test your knowledge and skills in
band structure concepts and calculations.
Send the report as a
single MS Word or pdf document to the instructor by email.
1. (3 pt) Use the orthogonal tight-binding code skband to calculate the
electronic band structure of a linear carbon chain.
2. (2 pt) Use the orthogonal tight-binding code skband to calculate the
electronic band structure of a dimerized linear carbon chain.
- Start by modifying the input file of problem 1.
- Keep the chain aligned along the x-direction in space.
- There will still be 2 atoms per unit cell. The new unit cell should be as long as
in problem 1, 2.6 Å.
- Use 1.2 Å and 1.4 Å as alternating interatomic distances.
- Use the same k-point sampling as in problem 1.
- Plot the band structure.
- Where is the Fermi level?
- Is the system metallic or insulating?
3. (6 pt) Use the orthogonal tight-binding code skband to calculate the
electronic band structure of graphyne, shown in the middle panel of the image below.
In comparison to graphene with two atoms per unit cell, graphyne has 12 atoms
per unit cell. Still, the shape of the unit cell and the symmetry of the Bravais
lattice are the same, so graphene and graphyne have the same set of k-points.
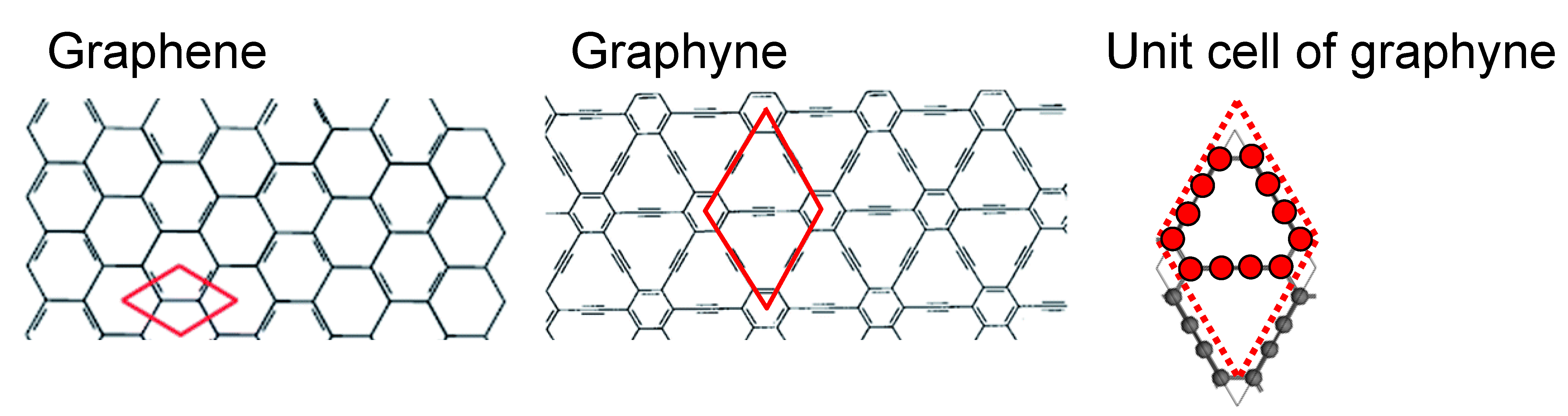
- Make a graphyne.xyz file with the coordinates of the 12 atoms per unit cell.
All interatomic distances should be 1.3 Å. Display the molecular structure
with Discovery Studio Visualizer. Label the atoms by their number in your solution.
- How long are the lattice constants? (Get the inspiration from the
C-gra-skin file).
- Generate the skin file for graphyne.
- Calculate and plot the band structure in the energy range between -5 eV and +5 eV
(you will need to modify the y-range called yr in skbnd.gpl).
- How many electrons are in the unit cell? How many bands are occupied?
- Where is the Fermi level?
- Is the system metallic, semimetallic, or insulating?
- Where (at which k-point) is the top of the valence band?
- What is the character of the state at the top of the valence band?
Return to the home page of PHY919
Document
http://www.pa.msu.edu/people/tomanek/PHY919/homework/PHY919HW6.html
has been visited
[an error occurred while processing this directive].
Last update:
2017.08.29 (Tuesday) 10:53:20 EDT.
by David Tomanek -
 tomanek@pa.msu.edu
tomanek@pa.msu.edu

