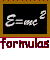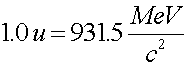Masses and energy According to the relation E = mc2, when the energy of a system changes, the mass changes as well. This is true for atomic, chemical or nuclear processes. Since nuclear processes release more energy per the mass of fuel involved, the association of Einstein and nuclear processes is stronger. In nuclear processes, the percentage of mass change is detectable, even though it is still only a fraction of a percent. In fact, the energy of nuclear states is often measured in mass units. One atomic mass unit (noted by a.u. or amu or u) corresponds to one twelth the mass of a carbon-12 nucleus.
When nuclei decay, they always go to a lower energy state. The excess energy not contained in nuclei or nucleons, the Q-value, is taken up by photons, neutrinos or electrons. By summing the before and after masses of a reaction, one can calculate the Q-value. |