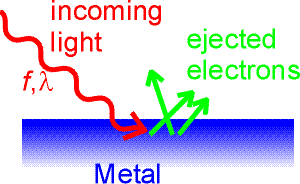
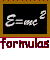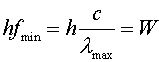The photoelectric effect In 1905 Einstein wrote three papers. The paper expounding the theory of special relativity did not win him the Nobel prize. Instead, it was his explanation of the photoelectric effect. The photoelectric effect refers to the interaction of monochromatic light with electrons in a metal. Electrons are bound to a metal, and can not escape the metal without overcoming a potential energy W, the work function of the metal. Typically, work functions are a few eV. Light incident on the metal might give the electrons enough energy to escape the metal. The electrons are then collected on an anode and measured as a current.
The interesting aspect of the photoelectric effect is that the current would stay practically zero for long-wavelength light, no matter what the intensity of the light. Einstein's explanation was that only photons of sufficiently high frequency (or sufficiently short wavelength) can give an electron enough energy to escape the metal. By adjusting the voltage on the metal, one can effectively control the work function, and by adjusting the wavelength of the incoming light, one can truly explore the expression E = hf. The current will begin only when the frequency of the incoming light corresponds to photons whose energy can overcome the work function.
The min and max refer to the minimum and maximum values to create the current. Photoelectric experiments allow one to determine Planck's constant in a controlled experiment. |